Home > Glossary of Key Terms
Glossary of Key Terms
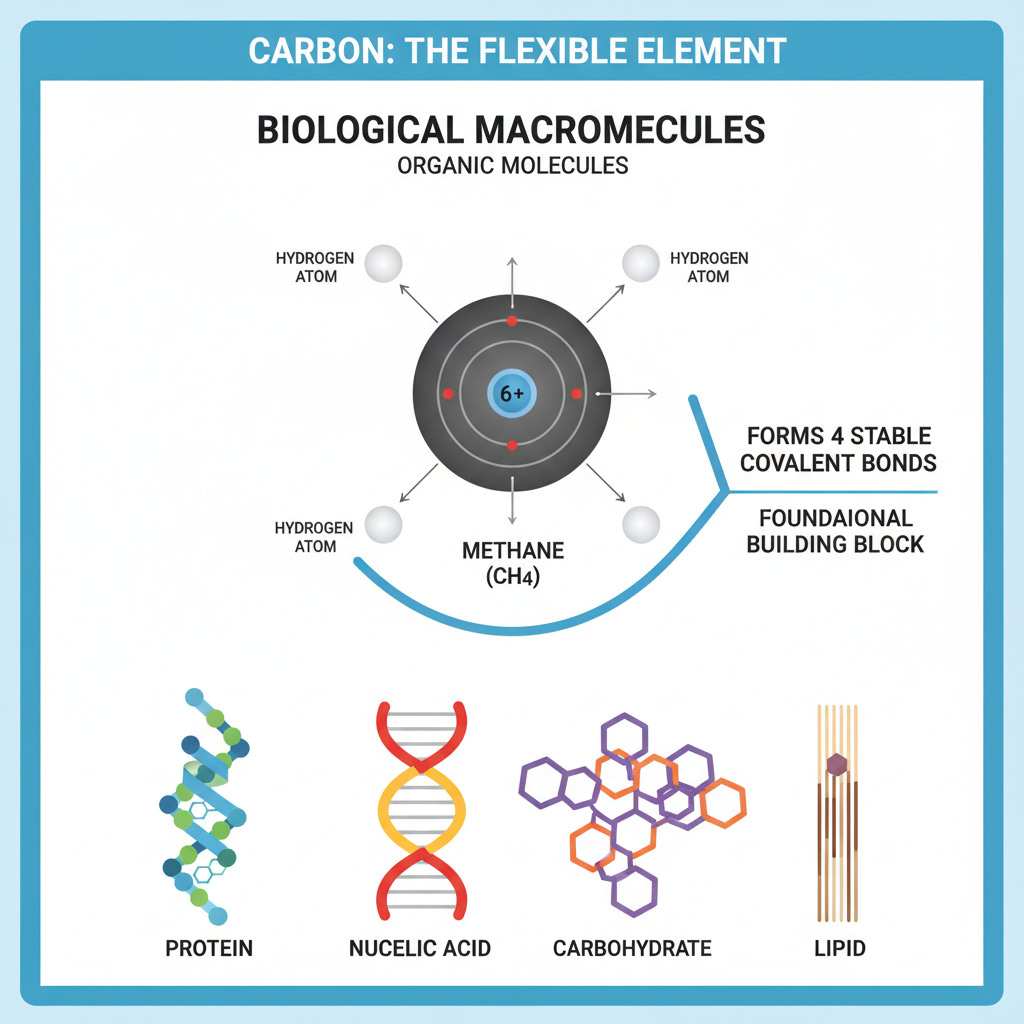
Macromolecules
Complex molecules, such as proteins, nucleic acids (like RNA and DNA), carbohydrates, and lipids, which are vital components of cells.
Organic molecule
Any substance, whether liquid, solid, or gas, that contains carbon, and is especially vital for life.
Carbon
The foundational element that serves as the main structural component, or 'backbone,' for all biological macromolecules.
Carbon atom structure
An atom with an atomic number of 6, possessing two electrons in its inner shell and four valence electrons in its second (outermost) shell.
Octet rule
The principle stating that atoms tend to form bonds so they have eight electrons in their outermost electron shell to achieve stability.
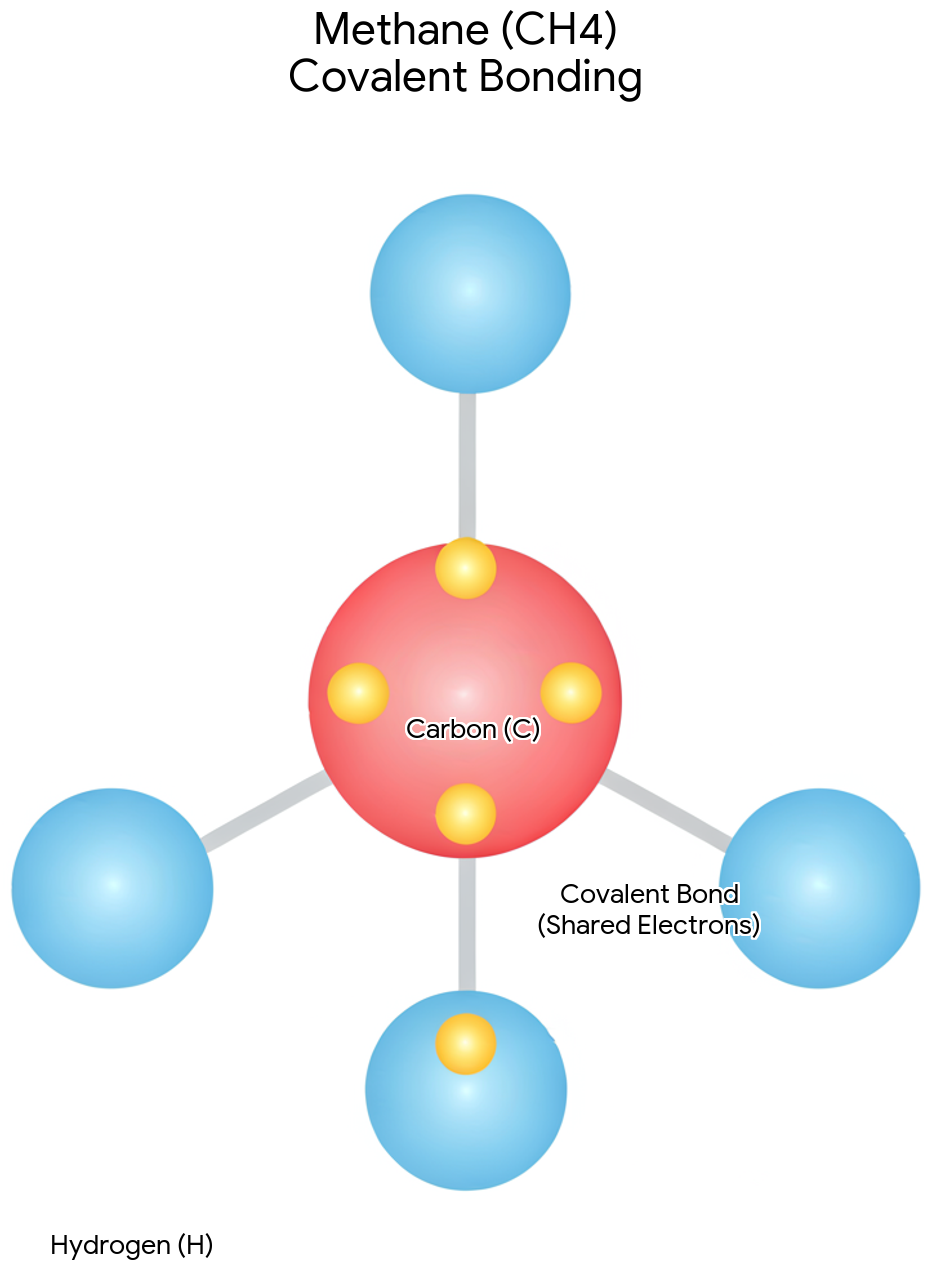
Covalent bonds
Chemical bonds formed by the sharing of electron pairs between atoms, which carbon atoms can form up to four of.
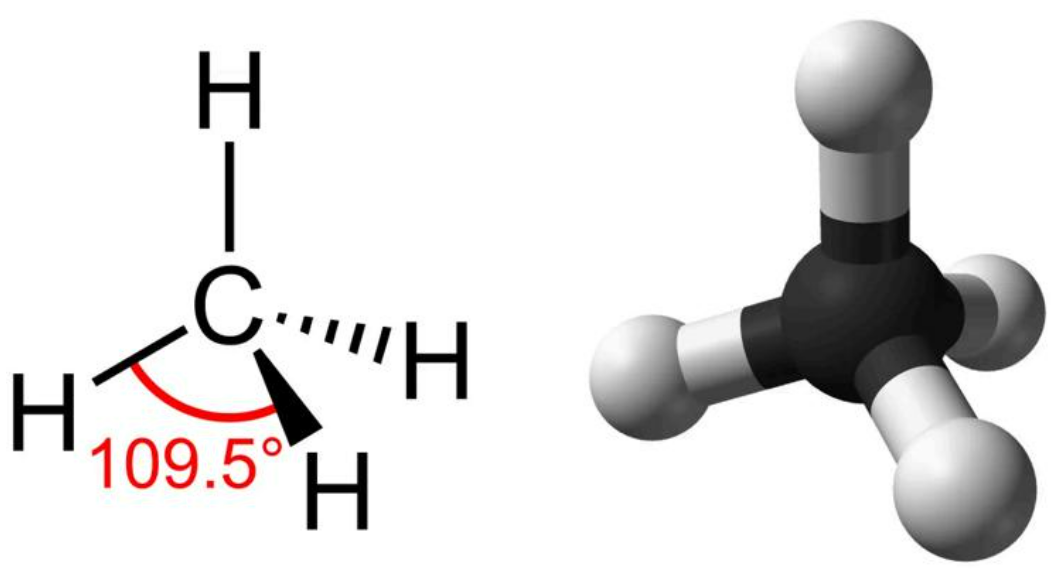
Methane (CH4)
The simplest hydrocarbon molecule, featuring a central carbon atom bonded by single covalent bonds to four different hydrogen atoms, resulting in a tetrahedral geometry.
Tetrahedral geometry
A three-dimensional molecular shape where a central carbon atom forms four single bonds, positioning the bonded atoms at the vertices of a tetrahedron with approximately 109.5° angles.
Hydrocarbons
Organic molecules made entirely of carbon and hydrogen, capable of storing large amounts of energy in their covalent bonds.
Hydrocarbon chains
A series of bonds between carbon atoms that can be straight, branched, or unbranched, forming the main structure of large molecules.
Single covalent bond
A carbon-carbon bond formed by sharing one pair of electrons, allowing rotation around its axis and leading to tetrahedral geometry around the carbon.
Double covalent bond
A carbon-carbon bond formed by sharing two pairs of electrons, resulting in a flat (planar) molecular arrangement and restricting rotation around the bond.
Triple covalent bond
A carbon-carbon bond formed by sharing three pairs of electrons, leading to a straight (linear) molecular arrangement and highly restricted rotation.
Planar arrangement
A molecular geometry where atoms, particularly those involved in a double bond, lie in a single flat plane.
Linear arrangement
A molecular geometry where atoms, particularly those involved in a triple bond, are arranged in a straight line.
Aliphatic hydrocarbons
Hydrocarbons characterized by linear chains of carbon atoms or rings where all carbon-carbon bonds within the ring are single bonds.
Aromatic hydrocarbons
Hydrocarbons consisting of closed rings of carbon atoms that have alternating single and double bonds, such as the benzene ring.
Benzene ring
A specific type of aromatic hydrocarbon, typically a six-membered carbon ring with alternating single and double bonds, found in various biological molecules and identified as a carcinogen.
Carcinogen
A substance that has the potential to cause cancer.
Conformation
The specific three-dimensional shape or spatial arrangement of a molecule, which is crucial to how large molecules (macromolecules) function.
See also: Summary & Key Points